Maintaining Data Integrity: Understanding FDA ALCOA Guidance
Did you know that lack of data integrity has been a top reason for FDA Drug GMP warning letters?*
In their 2018 Questions and Answers guidance on the implementation of their 21 CFR Part 11 data integrity rule, the FDA use the acronym ALCOA, where they define good data integrity practice as creating records that are attributable to the technician carrying out the testing, are legible, are created contemporaneously, original and accurate.

Attributable
Attributable can be interpreted to mean that records should include an electronic ‘signature’ to link them to the instrument/person that made the measurement and they should also include a reference to the water system being tested and the date and time it was taken. This implies electronic signatures for users signed on to the system. Control over electronic signature format can be site specific and is usually controlled by the IT department using Microsoft Active Directory controls. Ideally the on-line instrumentation should be configured to follow Active Directory controls to ensure correct electronic signature format according to the site-specific rules.
Legible records
The record of course is required to be legible, which implies that hand-written records are not acceptable. The FDA goes on to suggest that electronic records should be stored in a format that is open and can be read on many computing formats so that it will be accessible and readable for years to come. The FDA recommends typical formats such as PDF, XML or SGML.
Contemporaneous
The word contemporaneously implies that the electronic records should be created immediately when the sample is measured, implying that manual transcription of paper records is not good practice and that collating paper records and then manually transcribing them into electronic format at a later time or date is not good practice either.
Original
Naturally there is a danger with every transcription of test results from one form to another. Even scanning multiple paper records into electronic format runs the risk of duplication or missed scans. So the FDA recommends that the electronic record should be the original record created when the test was completed. Obviously, manually transcribed records are the riskiest, attracting the biggest opportunity for human error.
Accurate records
Finally the A in ALCOA. Naturally the electronic records should be accurate. This implies that the process for capturing those electronic records should be robust, i.e. manual calculations and manual data entry where opportunities for human error exist should be avoided. Electronic records generated by on-line instruments should contain information that links the data to the instrument used to make the measurement and the time and date of the measurement.
Our instruments are designed to facilitate ALCOA guidance. Discover the details here:
Products Designed for ALCOA Guidance
QC Step: Raw Material |
|||||||
Instrument |
Application |
Regulation |
A |
L |
C |
O |
A |
| ANATEL PAT700 TOC Analyzer | On-line Water for Injection (WFI), Purified Water (PW), Total Organic Carbon (TOC), Temperature and Conductivity | Multi-level, Individual User Name and Password for all users | Legible secure PDF export for Alarm Trail, Audit Trail, Measurement Results | Secure PDF created on day of sample analysis | Original electronic record created directly from the instrument | Original electronic record created directly from the instrument | |
QC Step: Production |
|||||||
Instrument |
Application |
Regulation |
A |
L |
C |
O |
A |
| Vi-CELL BLU Cell Viability Analyzer | Mammalian cell viability and concentration | USP <1046> | Multi-level, Individual User Name and Password for all users | Legible secure PDF export for measurement Results | Secure data records stored and tracked at time of measurement | Original electronic record created directly from the instrument | SOP parameters pre-programed into the instrument and automated |
| Vi-CELL MetaFLEX Bioprocessing Monitor | Mammalian cell bioreactor media health | Multi-level, Individual User Name and Password for all users | Legible reports can be printed or viewed on the instrument | Secure data records stored and tracked at time of measurement | Original electronic record created directly from the instrument | SOP parameters pre-programed into the instrument and automated |
|
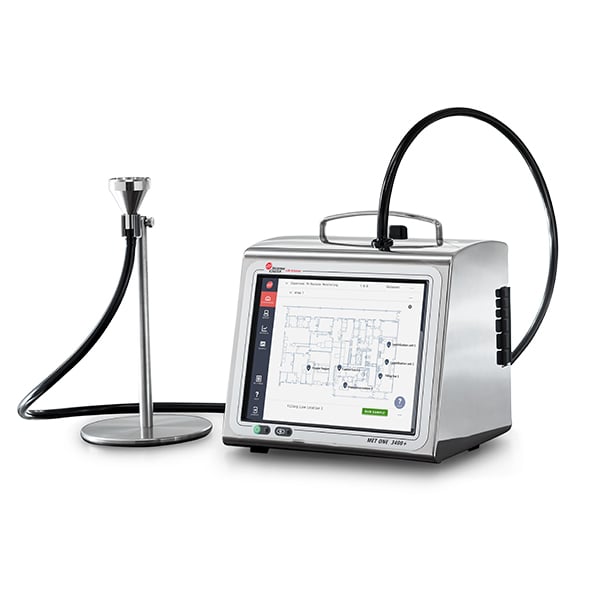
| MET ONE 3400+ Air Particle Monitor | Routine environmental monitoring (air particulates) in sterile manufacturing cleanrooms |
EU GMP Annex 1, CGMP, ISO 14644-1 & -2 | Multi-level, Microsoft Active Directory Username and Password control creates unique electronic signatures for users | Legible secure PDF export for Measurement Results including metadata | Secure PDF created on day of sample analysis | Original electronic record created directly from the instrument | SOP parameters pre-programed into the instrument and automated |
| MET ONE Facility Monitoring System | Sterile manufacturing cleanroom continuous air particulate monitoring | EU GMP Annex 1, CGMP, ISO 14644-2 | Multi-level, Individual User Name and Password for all users | Legible secure PDF export for Alarm Trail, Audit Trail, Measurement Results | Secure data records stored in database at time of measurement | Original electronic record created directly inside the counter | Your SOPs and sampling maps uploaded into the counter with version control. Completed records reviewed and approved for accuracy in the counter remotely via web-browser. No manual counter configuration or sample location entry. |
QC Step: Final Product Testing |
|||||||
Instrument |
Application |
Regulation |
A |
L |
C |
O |
A |
| HIAC 9703+ Liquid Particle Counter | Final product sub-visible particulate counting in parenteral drug products |
USP <788> USP <789> |
Multi-level, individual User Name and Password for all users | Legible, secure PDF export for Alarm Trail, Audit Trail, Measurement Results |
Secure PDF created on day of sample analysis | Original electronic record created directly from the instrument | SOP parameters pre-programed into the instrument and automated |
| Multisizer 4e Coulter Counter | Final product visible particulate counting in parenteral drug products |
Multi-level, individual, user name and password for all users |
Legible secure PDF export for Audit Trail, Measurement Results | Secure PDF created on day of sample analysis | Original electronic record created directly from the instrument | SOP parameters pre-programed into the instrument and automated | |
*Pharmaceutical Online, An Analysis Of FDA FY2018 Drug GMP Warning Letters By Barbara Unger, Unger Consulting Inc., https://www.pharmaceuticalonline.com/doc/an-analysis-of-fda-fy-druggmp-warning-letters-0003, published February 1, 2019
Reflection paper on expectations for electronic source data and data transcribed to electronic data collection tools in clinical trials By GCP Inspectors Working Group https://www.ema.europa.eu/en/documents/regulatory-procedural-guideline/reflection-paper-expectations-electronic-source-datadata- transcribed-electronic-data-collection_en.pdf, published June 09, 2010