The Discovery of EVs
Science and medicine have been at the forefront of some of the most fascinating discoveries in history. Take, for example, the charming story of penicillium mold accidentally falling into an agar plate filled with bacteria, leading to the serendipitous discovery of antibiotics. This discovery was a result of learning by chance. Another remarkable story is that of Barry Marshall, who bravely self-administered Helicobacter pylori to demonstrate its effect on peptic ulcers. This bold experiment ultimately earned him a Nobel Prize, showcasing the power of learning through experimentation.
We mustn't forget the extraordinary Marie Curie, who used to carry radium tubes in her pocket. Sadly, her exposure to radioactivity contributed to her developing leukemia, serving as a costly lesson learned by mistake. These anecdotes highlight the incredible journey of scientific curiosity.
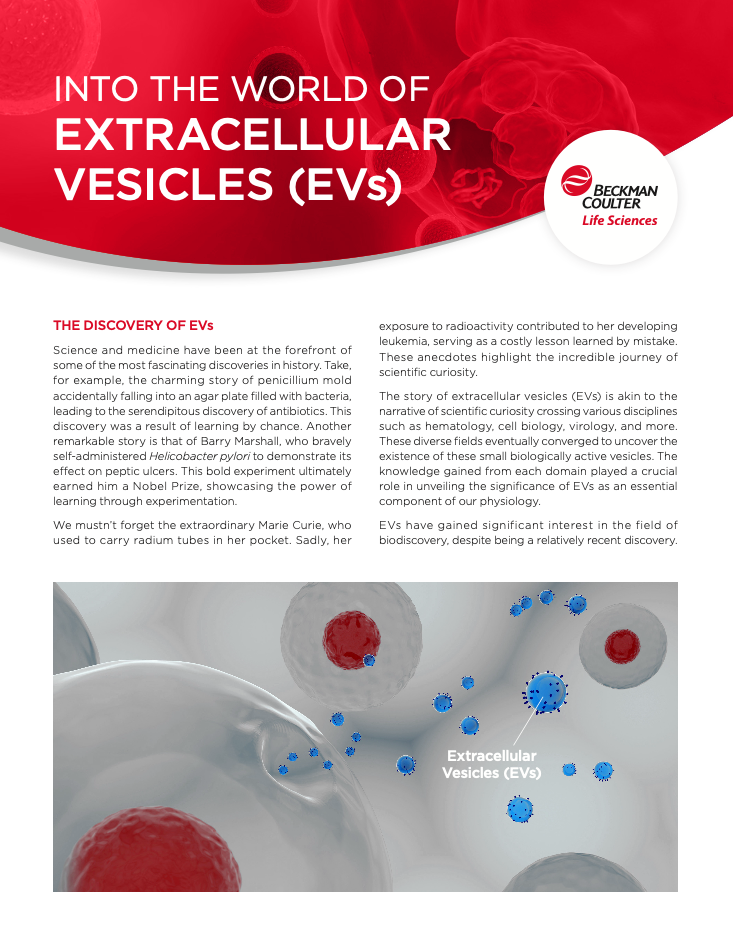
The story of extracellular vesicles (EVs) is akin to the narrative of scientific curiosity crossing various disciplines such as hematology, cell biology, virology, and more. These diverse fields eventually converged to uncover the existence of these small biologically active vesicles. The knowledge gained from each domain played a crucial role in unveiling the significance of EVs as an essential component of our physiology.
EVs have gained significant interest in the field of biodiscovery, despite being a relatively recent discovery. Many scientists are now focused on understanding the diversity of EVs, their role in physiological functions, and their potential involvement in various diseases.

In the 1930s, a biochemist named Chargaff was working on developing a centrifugation protocol to separate clotting factors from blood. During this process, he observed that adding the high-speed sediment to the supernatant plasma significantly shortened the clotting time (1). This observation brought to light the existence of EVs, a distinct family of biomolecules present in blood. With the assistance of West (2), they further identified a "particulate fraction" that sedimented at 31,000 x g and exhibited high clotting potential and thromboplastic properties. This fraction included EVs. Initially, it was hypothesized that these isolated fractions were composed of various minute breakdown products of blood corpuscles. However, it took nearly two more decades of research to identify and differentiate the material in minute particulate form, sedimentable by high-speed centrifugation, and originating from platelets but distinct from intact platelets. These findings align closely with the characteristics of EVs (3). The first electron microscopy images of this "platelet dust" provided scientists with visual insights into the true appearance of EVs (Figure 1).
EVs at a glance
- Extracellular vesicles (EVs) possess a phospholipid membrane that typically has a thickness of approximately 5 nm.
- These vesicles exhibit a wide range of sizes, with most fluids containing EVs of varying sizes.
- The minimum size of EVs is dependent on their composition and ranges from 30-50 nm in human blood plasma.
- Generally, the size distribution of EVs has a peak below 200 nm.
- In human blood, spherical EVs smaller than 500 nm constitute around 95% of the total population.
 |
Figure 1. Urinary EVs (uEVs) characterization by transmission electron microscopy (TEM). Representative picture of TEM characterization of uEVs isolated with ultracentrifugation. Bertolone et al. Front. Endocrinol. 2023 14:1096441. Published under a Creative Commons Attribution 4.0 International License: http://creativecommons.org/licenses/by/4.0/ |
The subsequent work by Crawford played a crucial role in identifying the presence of lipids and proteins within the membrane of EVs, shedding light on their functional properties (4). In a fascinating study on bat hibernation, scientists initially observed how EVs are generated from cells through the fusion of multivesicular bodies with the plasma membrane before being released (5). This discovery led to the realization that EVs are not exclusive to mammalian cells but are produced by various cell types (6). During the 1980s, researchers focused on unraveling the intricacies of membrane trafficking and the assembly of EVs, specifically examining how proteins are included or excluded (7).
Initially referred to as exosomes (small microvesicles), the term "extracellular vesicles" was officially adopted in 2011 to define these non-replicating structures enclosed by a lipid bilayer. The same year witnessed the establishment of the International Society of Extracellular Vesicles (ISEV), which developed guidelines (MISEV) to standardize the analysis and reporting of EV research (8). Since then, numerous topics have emerged, including the study of cargo trafficking, biomarkers, cancer, cardiovascular diseases, infectious diseases, drug delivery, and many more. These areas of research highlight the tremendous importance of EVs in various fields of study.
The Importance of EVs In Science and Medicine
The discovery of the world of extracellular vesicles (EVs) has brought to light previously unknown aspects of how the body functions, how cancer cells spread, and how pathogens disseminate. However, there is still much investigation needed to fully decipher the secrets of EVs. Scientists now recognize that EVs play a central role not only in physiological processes but also in pathological conditions. This recognition has led to a significant surge in research over the past decade, with thousands of scientific papers published in 2022 alone (Figure 2).
EVs are produced by all cell types across different life kingdoms. Their biogenesis can be categorized into two types of release: (i) budding from the plasma membrane, resulting in larger microvesicles and apoptotic bodies, and (ii) release of vesicles derived from endosomes (9). EVs are small in size (ranging from 30 to 500 nm) and possess an intraluminal region. Due to their size and ability to carry proteins and nucleic acids, scientists are focused on understanding their functions and exploring their potential as biomarkers.
Research has revealed that EVs play a role in various physiological processes, including angiogenesis, cellular migration, and cell-to-cell signaling. Their abundance, and their ability to easily enter target cells suggest their involvement in numerous physiological processes, both in peripheral circulation and tissues (Figure 3). In fact, EVs have the capacity to cross the blood-brain barrier, indicating a potential role in neurodegenerative diseases (10). Additionally, EVs can serve as biomarkers for peripheral screening tools in diseases like Alzheimer's (among other applications) (11).
There is growing interest in the therapeutic potential of EVs. Clinical trials are investigating the use of EVs as cargo vehicles to deliver compounds to target cells. By engineering EVs to express specific markers, they can be targeted to specific sites in the body and deliver cargo inside the cells. This targeted delivery offers advantages in terms of specificity compared to conventional drug delivery methods. Transmembrane proteins like tetraspanins (e.g., CD9, CD63, CD81), often used as markers of EVs, also play a role in this process (12). Once inside the cells, the cargo interacts with intracellular components, influencing signaling pathways and ultimately leading to desired outcomes such as activation, suppression, or modulation of cellular functions.
This constant physiological process, where EVs modulate cellular functions from within, impacts the interaction of target cells with their microenvironment, particularly in tissues. It is through the release of EVs that some cancer cells can spread to other tissues, as these vesicles have the ability to modulate the tissue microenvironment in favor of cancer cell dissemination (13). This area of research is highly active, as finding ways to suppress this action could potentially reduce the spread of cancer cells throughout the body (14).
How EVs Are Generated:
- EVs are derived from the endosomal system, leading to the formation of multivesicular bodies.
- These EVs carry cell-derived proteins on their surface.
- Subsequently, they are transported to the cell membrane and fuse with it.
- Once fused, they are released from the cell into the extracellular space.
- EVs can then bind to target cells that express the corresponding receptor that they themselves express.
- Once inside the target cell, they have the ability to release their cargo.

Figure 3: Biogenesis of vesicles from cells and their anchoring into target cells. EVs and their cargos are enclosed within microvesicular bodies (MVB) that migrate to the plasma membrane for release into the extracellular space.
Centrifugation At The Genesis of EV Discovery
The study of extracellular vesicles (EVs) and the development of the EV field have heavily relied on centrifugation protocols since Chargaff's initial discovery. Over the years, scientists have refined protocols to enrich, isolate, and characterize EVs. The ideal separation protocol would offer high yield, heterogeneity, and efficiency while minimizing time and costs. Centrifugation, being a well-established technique in laboratories, has been the method of choice that has propelled the field to its current state. There are various methods for isolating EVs, and the specific downstream applications dictate the appropriate protocol to employ, each with its own advantages and disadvantages (15). Body fluids such as plasma, serum, urine, cerebrospinal fluid, and milk are commonly used as starting materials for EV separation.
Ultracentrifugation, including differential centrifugation, is a widely employed isolation method for EVs. While a combination of techniques may be utilized, centrifugation is almost always included in the isolation protocols (Figure 4). Density gradient centrifugation, filtration, and size-exclusion chromatography have gained interest among researchers. However, subsequent purification methods still involve ultracentrifugation, density gradient centrifugation, or chromatography techniques. Magnetic bead separation, although offering a targeted approach, may compromise the true heterogeneity of EVs. The isolation of EVs entails the removal of contaminants and the separation of EVs from other particles, such as chylomicrons and lipids, present in biological fluids or cell culture media (Figure 4). Ultracentrifugation utilizes centrifugal force to separate components based on size and/or density. Differential ultracentrifugation techniques involve multiple spins at progressively increasing speeds to eliminate contaminants and pellet EVs. Density gradient ultracentrifugation is a high-resolution purification technique that separates particles based on physical properties (size, shape, mass, and/or density) by employing a medium with graded densities, such as sucrose or iodixanol. While density cushion ultracentrifugation strikes a balance between throughput and purity, density gradient ultracentrifugation provides the highest purity, and differential ultracentrifugation allows for the highest throughput. Density gradient and differential ultracentrifugation yield the cleanest samples and preserve the structural integrity of EVs compared to other methods like size exclusion chromatography or precipitation. The question of scalability, especially in the context of manufacturing and EV therapy, becomes important when deciding which isolation method to employ (16-19).

Figure 4: Isolating EVs involves the removal of contaminants and the separation of EVs from other particles, such as chylomicrons and lipids, present in biological fluids or cell culture media. Ultracentrifugation utilizes centrifugal force to separate components based on their size and/or density. Differential ultracentrifugation techniques employ multiple spins at progressively increasing speeds to first eliminate contaminants and then pellet EVs. Density gradient ultracentrifugation is a high-resolution purification technique that relies on a medium with graded densities, such as sucrose or iodixanol, to separate particles based on their physical properties such as size, shape, mass, and density.
Moving Forward With EV Research
From the initial discovery of EVs to the present day, several decades have passed. The recent excitement surrounding EVs stems from the potential applications that could arise from a deeper understanding of their generation, regulation, and manipulation for therapeutic purposes. Since EVs are produced by all types of living cells and have the ability to regulate numerous processes, the possibilities for their applications are limitless. However, this also emphasizes the need for further research to unravel the secrets of EVs. The scientific community is eager to standardize the isolation, characterization, and use of EVs, as this will enhance reproducibility and facilitate the translation of findings into clinical use.
There are still important characteristics of EVs that need to be clarified. Firstly, their heterogeneity is a significant aspect to explore. EVs are not a single entity, as their composition and size can vary even within the same cell. The generation and composition of EVs are likely to be continuously regulated processes influenced by the cell's status and received signals (Figure 5). Characterizing EVs individually, which can be achieved using technologies like flow cytometry, will contribute to a better understanding of their heterogeneity. Secondly, it is crucial to develop scalable methods for generating EVs in vitro for delivery to patients. Defining standard workflows will be essential in this regard, and centrifugation plays a crucial role in the manufacturing process. Lastly, mapping markers associated with different classes of EVs (e.g., cells of origin, healthy vs. cancer cells, activation status, disease biomarkers) is necessary to expedite ongoing therapeutic efforts.

Figure 5: Representation of the size and heterogeneity of EVs. High-density lipoproteins (HDLs), low-density lipoproteins (LDLs), and very low-density lipoproteins (vLDLs) are included for comparison. The average sizes of antibodies, viruses, and bacteria are also shown.
For Research Use Only. Not for use in diagnostic procedures.
Download the Application Note
The story of extracellular vesicles (EVs) is akin to the narrative of scientific curiosity crossing various disciplines such as hematology, cell biology, virology, and more. These diverse fields eventually converged to uncover the existence of these small biologically active vesicles. The knowledge gained from each domain played a crucial role in unveiling the significance of EVs as an essential component of our physiology.
Refrences
- Chargaff, E. J. (1945). Cell structure and the problem of blood coagulation. 160(1), 351–359.
- Chargaff, E., & West, R. (1946). The biological significance of the thromboplastic protein of blood. Journal of Biological Chemistry, 166(1), 189–197
- Wolf, P. (1967). The nature and significance of platelet products in human plasma. British Journal of Haematology, 13(3), 269–288
- Crawford, N. (1971). The presence of contractile proteins in platelet microparticles isolated from human and animal platelet-free plasma. British Journal of Haematology, 21(1), 53–69
- Nunez, E. A., Wallis, J., & Gershon, M. D. (1974). Secretory processes in follicular cells of the bat thyroid. 3. The occurrence of extracellular vesicles and colloid droplets during arousal from hibernation. American Journal of Anatomy, 141(2), 179–201
- Couch Y, Buzàs EI, Di Vizio D, Gho YS, Harrison P, Hill AF, Lötvall J, Raposo G, Stahl PD, Théry C, Witwer KW, Carter DRF. A brief history of nearly EV-erything - The rise and rise of extracellular vesicles. J Extracell Vesicles. 2021 Dec;10(14):e12144.
- Harding, C., Heuser, J., & Stahl, P. (1983). Receptor-mediated endocytosis of transferrin and recycling of the transferrin receptor in rat reticulocytes. Journal of Cell Biology, 97(2), 329–339
- Théry, C., Witwer, K. W., Aikawa, E., Alcaraz, M. J., Anderson, J. D., Andriantsitohaina, R., Antoniou, A., Arab, T., Archer, F., Atkin-Smith, G. K., Ayre, D. C., Bach, J.-M., Bachurski, D., Baharvand, H., Balaj, L., Baldacchino, S., Bauer, N. N., Baxter, A. A., Bebawy, M., & Zuba-Surma, E. K. (2018). Minimal information for studies of extracellular vesicles 2018 (MISEV2018): A position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. Journal of Extracellular Vesicles, 7(1), 1535750.
- Welsh JA, Arkesteijn GJA, Bremer M, Cimorelli M, Dignat-George F, Giebel B, Görgens A, Hendrix A, Kuiper M, Lacroix R, Lannigan J, van Leeuwen TG, Lozano-Andrés E, Rao S, Robert S, de Rond L, Tang VA, Tertel T, Yan X, Wauben MHM, Nolan JP, Jones JC, Nieuwland R, van der Pol E. A compendium of single extracellular vesicle flow cytometry. J Extracell Vesicles. 2023 Feb;12(2):e12299. doi: 10.1002/jev2.12299. PMID: 36759917; PMCID: PMC9911638.
- Jeyaraman M, Rajendran RL, Muthu S, Jeyaraman N, Sharma S, Jha SK, Muthukanagaraj P, Hong CM, Furtado da Fonseca L, Santos Duarte Lana JF, Ahn BC, Gangadaran P. An update on stem cell and stem cell-derived extracellular vesicle-based therapy in the management of Alzheimer’s disease. Heliyon. 2023 Jun 29;9(7):e17808. doi: 10.1016/j.heliyon.2023.e17808. PMID: 37449130; PMCID: PMC10336689.
- Cheng, L., Doecke, J., Sharples, R. et al. Prognostic serum miRNA biomarkers associated with Alzheimer’s disease shows concordance with neuropsychological and neuroimaging assessment. Mol Psychiatry 20, 1188–1196 (2015)
- Ginini L, Billan S, Fridman E, Gil Z. Insight into Extracellular Vesicle-Cell Communication: From Cell Recognition to Intracellular Fate. Cells. 2022 Apr 19;11(9):1375. doi: 10.3390/cells11091375. PMID: 35563681; PMCID: PMC9101098.
- Kosaka N, Yoshioka Y, Fujita Y, Ochiya T. Versatile Roles of Extracellular Vesicles in Cancer. J Clin Invest (2016) 126:1163–72.
- Xu Y, Yao Y, Yu L, Zhang X, Mao X, Tey SK, Wong SWK, Yeung CLS, Ng TH, Wong MYM, Che CM, Lee TKW, Gao Y, Cui Y, Yam JWP. Clathrin light chain A-enriched small extracellular vesicles remodel microvascular niche to induce hepatocellular carcinoma metastasis. J Extracell Vesicles. 2023 Aug;12(8):12359
- Gardiner, C., Vizio, D. D., Sahoo, S., Théry, C., Witwer, K. W., Wauben, M., & Hill, A. F. (2016). Techniques used for the isolation and characterization of extracellular vesicles: Results of a worldwide survey. Journal of Extracellular Vesicles, 5, 32945
- Dong X, Dong JF, Zhang J. Roles and therapeutic potential of different extracellular vesicle subtypes on traumatic brain injury. Cell Commun Signal. 2023 Aug 18;21(1):211
- García-Fernández J, Fuente Freire M. Exosome-like systems: Nanotechnology to overcome challenges for targeted cancer therapies. Cancer Lett. 2023 May 1;561:216151. doi: 10.1016/j.canlet.2023.216151.
-
Li G, Chen T, Dahlman J, Eniola-Adefeso L, Ghiran IC, Kurre P, Lam WA, Lang JK, Marbán E, Martín P, Momma S, Moos M, Nelson DJ, Raffai RL, Ren X, Sluijter JPG, Stott SL, Vunjak-Novakovic G, Walker ND, Wang Z, Witwer KW, Yang PC, Lundberg MS, Ochocinska MJ, Wong R, Zhou G, Chan SY, Das S, Sundd P. Current challenges and future directions for engineering extracellular vesicles for heart, lung, blood and sleep diseases. J Extracell Vesicles. 2023 Feb;12(2):e12305. doi: 10.1002/jev2.12305. Erratum in: J Extracell Vesicles. 2023 Mar;12(3):e12314. PMID: 36775986
-
Estes S, Konstantinov K, Young JD. Manufactured extracellular vesicles as human therapeutics: challenges, advances, and opportunities. Curr Opin Biotechnol. 2022 Oct;77:10277
Interested in learning how to advance your EV research?
Whether you have questions or are interested in a demo, we're here to help.