Error Prevention at Start Up, Cleaning and Worklist Generation
The AQUIOS CL system employs several features that work to prevent errors during startup, cleaning, and worklist generation. These prevention methods can be broken down into the following:
Table 2. Error Prevention Startup/Cleaning/Worklist Generation at a Glance
Autocleaning
Autocleaning is a function integrated directly into the AQUIOS system. The AQUIOS CL flow cytometer continually monitors and detects potential clogs and/or air bubbles in the flow cell. Upon detection of a potential clog or bubble in the flow cell, the system aborts the sample run and activates an autocleaning cycle. Once this cycle is complete, the system will rerun the aborted sample. Autocleaning reduces the potential for low precision and erroneous results. In addition, autocleaning reduces the need for sample reruns.
Test Requests to and from the LIS
Traditional flow cytometers do not automatically transfer test requests to and from the LIS without the added expense of middleware. If the LIS connection is enabled on the AQUIOS CL flow cytometer, the system automatically retrieves the test request from the LIS. Users with Reviewer access are authorized to review and transmit samples to the LIS. Reviewer users are able to generate and perform test requests, and review and release results.
Not only does the built-in LIS ensure automatic transfer of test requests reducing transcription errors, but no middleware is required. Different user access levels ensure that operators can only work on areas that align with their level of experience with the system.
Creating Worklists
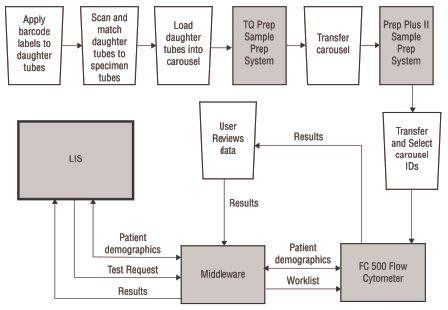
On a traditional flow cytometer, there is no direct connection from the worklist to sample preparation system, or sample preparation system to the analyzer. Traditional flow cytometers automatically send the results from the analyzer to the LIS and the test request from the middleware to the analyzer when using middleware. Potential errors may result because everything is not linked together. Refer to Figure 1 for a visual representation of an LIS connection using the FC 500 flow cytometer and TQ-Prep sample prep system.
Figure 1. LIS Connection for FC 500 Flow Cytometer and TQ-Prep Sample Prep System
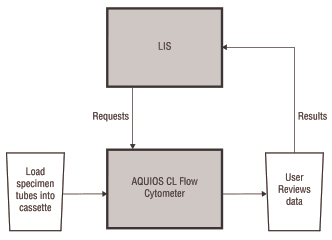
Conversely, the AQUIOS CL flow cytometer is one seamless system; no middleware is required. However, middleware can be used if desired. The system is able to send test requests to the analyzer and capture the information to automatically generate a worklist. Refer to Figure 2 for a visual representation of the LIS connection for the AQUIOS CL flow cytometer.
Figure 2. LIS Connection for the AQUIOS CL Flow Cytometer
Fill the form to download Workflow Comparison Study with AQUIOS CL Flow Cytometer

HIV Advanced Disease Management

HIV Advanced Disease Management Solutions
Monitoring CD4 lymphocyte counts is essential in providing critical information that impacts patient care. CD4 monitoring allows caregivers to know when the disease transforms and stage its progression so they can implement the most appropriate intervention..
Fast Throughput Subset Analysis with AQUIOS CL
In the clinical management of immune deficiency diseases, accurately counting the absolute cell numbers of leukocyte subsets and measuring the percentage of individual subtypes in blood is critical.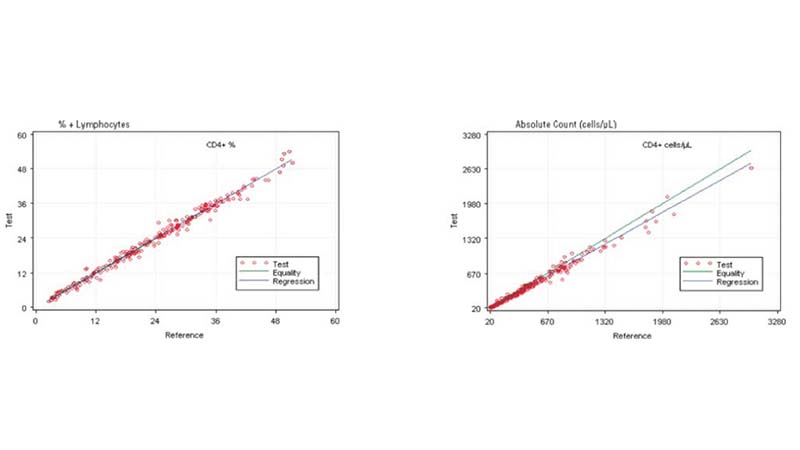
Comparing the AQUIOS CL PLG Application to the FC500 MCL FlowCARE PLG Application
Based on this study, the data suggests a strong correlation between the AQUIOS CL PLG application and the FC500 MCL FlowCARE PLG application.
AQUIOS CL Tetra and Aged Whole Blood samples
Based on this study, the AQUIOS CL instrument with Tetra application provides accurate results for recovery of the T, B and NK lymphocyte subsets in clinical and normal specimens collected into the EDTA K3 tubes when stored at room temperature for up to 24 hours.Error Prevention on AQUIOS CL overview
Laboratory standards and regulations set forth by governmental bodies such as the Centers for Disease Control (CDC) help to define the procedures that laboratories follow
Error prevention during Startup and Worklist creation
The AQUIOS CL system employs several features that work to prevent errors during startup, cleaning, and worklist generation, including Autocleaning, Test Requests to and from the LIS and Creating Worklists.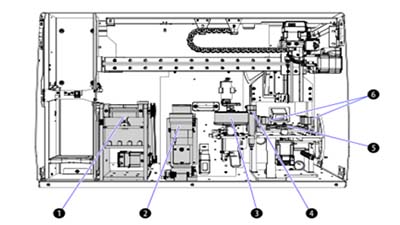
Error Prevention During Sample Prep
The AQUIOS CL system employs several features that work to prevent errors during sample prep.
Error Prevention During Quality Control
The AQUIOS CL system employs several features that work to prevent errors during quality control, including Automatically Pause when QC Fails, QC Checks, Daily QC (Controls), Real Time QC (Patient Sample).References:
- 1997 Revised Guidelines for the Performance of CD4+ T-Cell Determination in Persons with Human Immunodeficiency Virus (HIV) Infection. MMWR, January 10, 1997, p. 11.
- Levey S., Jennings, E.R., (1950) The use of control charts in the clinical laboratory. Am. J. Clin. Pathol. 20:1059.
- Hulspas R, Dombkowski D, Preffer F, Douglas D, Kildew-Shah B, Gilbert J. Flow Cytometry and the Stability of Phycoerythrin-Tandem Dye Conjugates. International Society for Advancement of Cytometry. September 23, 2009.
- Hulspas R, O’Gorman MRG, Wood BL, Gratama, JW, Sutherland DR. Considerations for the control of background fluorescence in clinical flow cytometry. Cytometry Part B 2009;76B:355-364.
- Centers for Disease Control and Prevention. 2003 Guidelines for performing single-platform absolute CD4+ T-cell determinations with CD45 gating for persons infected with Human Immunodeficiency Virus HIV). MMWR 2003; 57 (No. RR. 2): 1-13.